If Neanderthals ever walk the earth again, the primordial ooze from which they will rise is an emulsion of oil, water, and DNA capture beads engineered in the laboratory of 454 Life Sciences in Branford, Connecticut. Over the past 4 years those beads have been gathering tiny fragments of DNA from samples of dissolved organic materials, including pieces of Neanderthal bone. Genetic sequences have given paleoanthropologists a new line of evidence for testing ideas about the biology of our closest extinct relative.
The first studies of Neanderthal DNA focused on the genetic sequences of mitochondria, the microscopic organelles that convert food to energy within cells. In 2005, however, 454 began a collaborative project with the Max Planck Institute in Leipzig, Germany, to sequence the full genetic code of a Neanderthal woman who died in Croatia's Vindija cave 30,000 years ago. As the Neanderthal genome is painstakingly sequenced, the archaeologists and biologists who study it will be faced with an opportunity that seemed like science fiction just 10 years ago. They will be able to look at the genetic blueprint of humankind's nearest relative and understand its biology as intimately as our own.
In addition to giving scientists the ability to answer questions about Neanderthals' relationship to our own species – did we interbreed, are we separate species, who was smarter – the Neanderthal genome may be useful in researching medical treatments. Newly developed techniques could make cloning Neanderthal cells or body parts a reality within a few years. The ability to use the genes of extinct hominins is going to force the field of paleoanthropology into some unfamiliar ethical territory. There are still technical obstacles, but soon it could be possible to use that long-extinct genome to safely create a healthy, living Neanderthal clone. Should it be done?
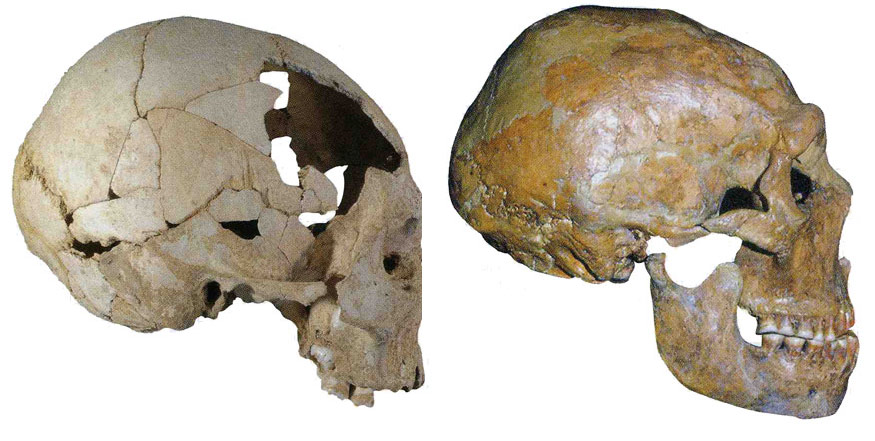
The 50,000-year-old skull of a Neanderthal from the site of Shanidar in Iran (top) has a prominent browridge and more projecting face than the 40,000-year-old Homo sapiens skull found at Pestera cu Oase in Romania. (Erik Trinkaus)
At the 454 Life Sciences offices, Gerald Irzyk, Jason Affourtit, and Thomas Jarvie explain the process they use to read the chemicals that made up Neanderthal DNA and the genes that determined a large part of their biology. DNA has a shape, called a double helix, that makes it look like a twisted ladder. Each rung on the ladder is called a base-pair. The rungs are made up of a pair of chemicals called nucleotides--adenine, thymine, cytosine, and guanine, which are usually referred to by their first initials. The sequence of the nucleotides in the DNA determines what genes an organism has and how they function.
Although most of the Neanderthal genome sequencing is now being done by the San Diego-based company Illumina, the Max Planck Institute initially chose 454 because it had come up with a way to read hundreds of thousands of DNA sequences at a time. Genome-sequencing technology is advancing at a rate comparable to computer processing power. "Six years ago if you wanted to sequence E. coli [a species of bacteria], which is about 4 million base-pairs in length, it would have taken one or maybe two million dollars, and it would have taken a year and 150 people," says Jarvie. "Nowadays, one person can do it in two days and it would cost a few hundred dollars."
Putting the fragments themselves in order can be a little tricky. "At first glance, it's just this completely random assemblage of As, Ts, Cs, and Gs," says Irzyk. "But it turns out there are patterns and motifs, and sometimes these are very specific to a group of organisms." For the Neanderthal sample, the human and chimpanzee genomes were used as references for checking the sequence.
Working with ancient DNA can be much more problematic than sequencing genetic material from living species. Within hours of death, cells begin to break down in a process called apoptosis. The dying cells release enzymes that chop up DNA into tiny pieces. In a human cell, this means that the entire three-billion-base-pair genome is reduced to fragments a few hundred base-pairs long or shorter. The DNA also goes through chemical changes that alter the nucleotides as it ages--C changes into T, and G turns into A--which can cause the gene sequence to be interpreted incorrectly. In the case of the Neanderthal sample, somewhere between 90 and 99 percent of the DNA came from bacteria and other contaminants that had found their way into the bone as it sat in the ground and in storage. The contaminant DNA has to be identified and eliminated. Given the similarity between Neanderthal and modern human DNA, this can be especially difficult when the contamination comes from the people who excavated or analyzed the bone.

Neanderthals and Homo sapiens were in contact for 5,000 to 7,000 years. Both lived in the same type of environments, but our species had a competitive advantage that gradually forced Neanderthals to the edges of Europe. These maps show the range of Neanderthals (left) and Homo sapiens (right) between 38,600 and 36,500 years ago. By this time, Neanderthals had been crowded out of Europe except the Iberian peninsula. The orange and gray areas were suitable for habitation. The white circles are archaeological sites. (Courtesy William Banks)
According to Stephan Schuster, a geneticist at the Pennsylvania State University, the first draft of the Neanderthal genome is likely to contain many errors. He estimates that getting a completely accurate DNA sequence will require taking five separate samples from the same individual, and sequencing that genome 30 times.
Schuster sequenced the mammoth genome in 2007, and that approach might work for large animals, but taking five samples from a single Neanderthal would require the destruction of a large amount of valuable bone. Carles Lalueza-Fox, a paleogeneticist at Spain's University of Barcelona, believes the accuracy of the DNA could be checked by resequencing dozens or hundreds of times the areas of the Neanderthal genome that seem likely to have errors.
Cloning a Neanderthal will take a lot more than just an accurately reconstructed genome. Artificially assembling an exact copy of the Neanderthal DNA sequence could be done easily and cheaply with current technology, but a free-floating strand of DNA isn't much good to a cell. "The bigger challenge is--how do you assemble a genome without a cell?" asks James Noonan, a geneticist at Yale University. "How do you package DNA into chromosomes, and get that into a nucleus? We don't know how to do that." The shape of the DNA within the chromosomes affects the way that genes interact with chemicals inside the cell. Those interactions control when, how much, and what types of proteins a cell's DNA produces. Those proteins are the building blocks of an organism, so the way a genome expresses itself is as important as the DNA. According to Schuster and Lalueza-Fox, the cellular damage that occurs after death makes it impossible to understand Neanderthal gene expression. This could mean that making a clone identical to someone who lived 30,000 years ago is impossible.
One way to get around the problems of working with an artificial genome would be to alter the DNA inside a living cell. This kind of genetic engineering can already be done, but very few changes can be made at one time. To clone a Neanderthal, thousands or possibly millions of changes would have to be made to a human cell's DNA. George Church, a professor of genetics at Harvard Medical School, is part of a research team that is developing a technique to make hundreds of alterations to a genome at the same time. The technique, multiplex automated genome engineering (MAGE), uses short strands of DNA called oligonucleotides to insert pieces of artificial genetic material into a cell's genome at specifically targeted sites. MAGE has been used successfully to make 24 alterations to the genomes of bacteria, mice, and, more recently, human cells. Church estimates that it would take about 10 million changes to make a modern human genome match the Neanderthal genome. Accomplishing this would be a matter of drastically scaling up the technique.
Church believes the place to start with Neanderthal cloning is on the cellular level, creating liver, pancreas, or brain cells. "You can't really tell anything from just looking at the gene sequence," he says. "It's hard to predict physical traits; you have to test them in living cells." Neanderthal cells could be important for discovering treatments to diseases that are largely human-specific, such as HIV, polio, and smallpox, he says. If Neanderthals are sufficiently different from modern humans, they may have a genetic immunity to these diseases. There may also be differences in their biology that lead to new drugs or gene therapy treatments.
So far, efforts to revive extinct species using cloning have a dismal track record. On January 6, 2000, a violent storm in northern Spain caused a tree branch to fall on Celia, the last Pyrenean Ibex, crushing her skull. That would seem like a clear indication that the ibex's evolutionary luck had run out, but a tissue sample taken from Celia's ear provided DNA that a team of Spanish scientists used to reconstruct 439 eggs. Only 57 developed into embryos, 52 did not survive the full term of the pregnancy, four were stillborn, and the one clone that survived birth died of lung failure within hours of delivery.
The ibex clones were created using techniques pioneered by Advanced Cell Technology, a biotechnology company in Worcester, Massachusetts. The technique, called nuclear transfer, involves removing the nucleus, the part containing the cell's genetic material, of a donor egg cell and replacing it with a nucleus containing clone DNA. In the ibex's case, goat eggs were used because the species are closely related and goats have been successfully cloned many times, explains Robert Lanza, ACT's chief scientific officer. According to Lanza, species such as cows and goats are now routinely cloned with few problems.
Species that have not been repeatedly cloned still face risks. The nuclear transfer process disrupts the cell and often causes it to die. The number of sick and dead individuals produced by nuclear transfer cloning is the reason nearly all scientists are opposed to human reproductive cloning. But even if nuclear transfer cloning could be perfected in humans or Neanderthals, it would likely require a horrifying period of trial and error. There is, however, another option.
The best way to clone Neanderthals may be to create stem cells that have their DNA. In recent years, geneticists have learned how to take skin cells and return them to a state called pluripotency, where they can become almost any type of cell in the human body. Church proposes to use the MAGE technique to alter a stem cell's DNA to match the Neanderthal genome. That stem cell would be left to reproduce, creating a colony of cells that could be programmed to become any type of cell that existed in the Neanderthal's body. Colonies of heart, brain, and liver cells, or possibly entire organs, could be grown for research purposes.
This technique could also be used to create a person. A stem cell with Neanderthal DNA could be implanted in a human blastocyst--a cluster of cells in the process of developing into an embryo. Then, all of the non-Neanderthal cells could be kept from growing. The individual who developed from that blastocyst would be entirely the result of Neanderthal genes. In effect, it would be a cloned Neanderthal. Church believes that after the earliest stages of development, the genes would express themselves as they did in the original individual, eliminating any influences from the modern human or chimpanzee cell.
The technique is new, and has only been tested in mice so far, but Church thinks it might work in humans. However, he points out that anyone cloned by this process would still be lacking the environmental and cultural factors that would have influenced how the original Neanderthals grew up. "They would be something new," Church says, "neo-Neanderthals."
In northern Spain 49,000 years ago, 11 Neanderthals were murdered. Their tooth enamel shows that each of them had gone through several periods of severe starvation, a condition their assailants probably shared. Cut marks on the bones indicate the people were butchered with stone tools. About 700 feet inside El Sidrön cave, a research team including Lalueza-Fox excavated 1,700 bones from that cannibalistic feast. Much of what is known about Neanderthal genetics comes from those 11 individuals.
Lalueza-Fox does not plan to sequence the entire genome of the El Sidrön Neanderthals. He is interested in specific genes. "I choose genes that are somehow related to individuality," he says. "I'd like to create a personal image of these guys." So far, his work has shown that Neanderthals had a unique variant of the gene for pale skin and red hair, which may mean their skin and hair color differed from modern humans. Lalueza-Fox tested the blood types of two Neanderthals and found they were both type O. He also discovered that modern humans and Neanderthals share a version of a gene called FOXP2, which is associated with language ability and means that Neanderthals probably spoke their own languages.
The Neanderthals broke away from the lineage of modern humans around 450,000 years ago. They evolved larger brains and became shorter than their likely ancestor, Homo heidelbergensis. They also developed a wider variety of stone tools and more efficient techniques for making them. On average, Neanderthals had brains that were 100 cubic centimeters (about 3 ounces) larger than those of people living today. But those differences are likely due to their larger overall body size. Those large brains were housed inside skulls that were broader and flatter, with lower foreheads than modern humans. Their faces protruded forward and lacked chins. Their arms and the lower part of their legs were shorter than modern humans', making them slower and less efficient runners, but they also had more muscle mass. Their bones were often thicker and stronger than ours, but they typically show a lot of healed breaks that are thought to result from hunting techniques requiring close contact with large game such as bison and mammoths. They had barrel-shaped chests and broad, projecting noses, traits some paleoanthropologists believe would have helped Neanderthals breathe more easily when chasing prey in cold environments.
Recent studies comparing Neanderthal and modern human anatomy have created some surprising insights. "Neanderthals are not just sort of funny Eskimos who lived 60,000 years ago," says Jean-Jacques Hublin, a paleoanthropologist at Max Planck. "They have a different way to give birth to babies, differences in life history, shape of inner ear, genetics, the speed of development of individuals, weaning, age of puberty." A study comparing Neanderthal and modern children showed Neanderthals had shorter childhoods. Some paleoanthropologists believe they reached physical maturity at age 15.
As different as Neanderthals were, they may not have been different enough to be considered a separate species. "There are humans today who are more different from each other in phenotype [physical characteristics]," says John Hawks, a paleoanthropologist at the University of Wisconsin. He has studied differences in the DNA of modern human populations to understand the rate of evolutionary change in Homo sapiens. Many of the differences between a Neanderthal clone and a modern human would be due to genetic changes our species has undergone since Neanderthals became extinct. "In the last 30,000 years we count about 2,500 to 3,000 events that resulted in positive functional changes [in the human genome]," says Hawks. Modern humans, he says, are as different from Homo sapiens who lived in the Neolithic period 10,000 years ago, as Neolithic people would have been from Neanderthals.
Clones created from a genome that is more than 30,000 years old will not have immunity to a wide variety of diseases, some of which would likely be fatal. They will be lactose intolerant, have difficulty metabolizing alcohol, be prone to developing Alzheimer's disease, and maybe most importantly, will have brains different from modern people's.
Bruce Lahn at the University of Chicago studies the evolutionary history of the genes that control human brain development. One gene that affects brain size particularly interests him, a variant of the microcephalin gene, which Lahn thinks may have entered the human gene pool through interbreeding with Neanderthals. If that turns out to be true, roughly 75 percent of the world's population has a brain gene inherited from Neanderthals. Lahn is excited to see what the Neanderthal microcephalin gene sequence looks like. "Is the Neanderthal sequence more similar to the ancestral version or the newer, derived version of the gene?" Lahn asks. "Or is the Neanderthal yet a third version that is very different from either of the two human versions? No matter how you look at it, it makes that data very interesting."
The Neanderthals' brains made them capable of some impressive cultural innovations. They were burying their dead as early as 110,000 years ago, which means that they had a social system that required formal disposal of the deceased. Around 40,000 years ago, they adopted new stone-tool-making traditions, the Châttelperronian tradition in Western Europe and the Uluzzian in Italy, that included a greater variety of tools than they had used in hundreds of thousands of years. But even if they were as adaptable as Homo sapiens, the question remains--if they were so smart, why are they dead? Chris Stringer of London's Natural History Museum believes our species hunted and gathered food so intensively that there simply was not enough room for the Neanderthals to make a living. In other words, they had the same problem as many species facing extinction today--they were crowded out of their ecological niche by Homo sapiens. Finding a place in the world for a Neanderthal clone would be only one dilemma that would have to be solved.
Bernard Rollin, a bioethicist and professor of philosophy at Colorado State University, doesn't believe that creating a Neanderthal clone would be an ethical problem in and of itself. The problem lies in how that individual would be treated by others. "I don't think it is fair to put people...into a circumstance where they are going to be mocked and possibly feared," he says, "and this is equally important, it's not going to have a peer group. Given that humans are at some level social beings, it would be grossly unfair." The sentiment was echoed by Stringer, "You would be bringing this Neanderthal back into a world it did not belong to....It doesn't have its home environment anymore."
There were no cities when the Neanderthals went extinct, and at their population's peak there may have only been 10,000 of them spread across Europe. A cloned Neanderthal might be missing the genetic adaptations we have evolved to cope with the world's greater population density, whatever those adaptations might be. But, not everyone agrees that Neanderthals were so different from modern humans that they would automatically be shunned as outcasts.
"I'm convinced that if one were to raise a Neanderthal in a modern human family he would function just like everybody else," says Trenton Holliday, a paleoanthropologist at Tulane University. "I have no reason to doubt he could speak and do all the things that modern humans do."
"I think there would be no question that if you cloned a Neanderthal, that individual would be recognized as having human rights under the Constitution and international treaties," says Lori Andrews, a professor at Chicago-Kent College of Law. The law does not define what a human being is, but legal scholars are debating questions of human rights in cases involving genetic engineering. "This is a species-altering event," says Andrews, "it changes the way we are creating a new generation." How much does a human genome need to be changed before the individual created from it is no longer considered human?
Legal precedent in the United States seems to be on the side of Neanderthal human rights. In 1997, Stuart Newman, a biology professor at New York Medical School attempted to patent the genome of a chimpanzee-human hybrid as a means of preventing anyone from creating such a creature. The patent office, however, turned down his application on the basis that it would violate the Constitution's 13th amendment prohibition against slavery. Andrews believes the patent office's ruling shows the law recognizes that an individual with a half-chimpanzee and half-human genome would deserve human rights. A Neanderthal would have a genome that is even more recognizably human than Newman's hybrid. "If we are going to give the Neanderthals humans rights...what's going to happen to that individual?" Andrews says. "Obviously, it won't have traditional freedoms. It's going to be studied and it's going to be experimented on. And yet, if it is accorded legal protections, it will have the right to not be the subject of research, so the very reasons for which you would create it would be an abridgment of rights."
Human rights laws vary widely around the world. "There is not a universal ban on cloning," says Anderson. "Even in the United States there are some states that ban it, others that don't." On August 8, 2005, the United Nations voted to ban human cloning. It sent a clear message that most governments believe that human cloning is unethical. The ban, however, is non-binding.
The legal issues surrounding a cloned Neanderthal would not stop with its rights. Under current laws, genomes can be patented, meaning that someone or some company could potentially own the genetic code of a long-dead person. Svante Pääbo, who heads the Neanderthal genome sequencing project at Max Planck, refused to comment for this article, citing concerns about violating an embargo agreement with the journal that is going to publish the genome sequence. But he did send ARCHAEOLOGY this statement: "We have no plans to patent any of the genes in the Neanderthal."
The ultimate goal of studying human evolution is to better understand the human race. The opportunity to meet a Neanderthal and see firsthand our common but separate humanity seems, on the surface, too good to pass up. But what if the thing we learned from cloning a Neanderthal is that our curiosity is greater than our compassion? Would there be enough scientific benefit to make it worth the risks? "I'd rather not be on record saying there would," Holliday told me, laughing at the question. "I mean, come on, of course I'd like to see a cloned Neanderthal, but my desire to see a cloned Neanderthal and the little bit of information we would get out of it...I don't think it would be worth the obvious problems." Hublin takes a harder line. "We are not Frankenstein doctors who use human genes to create creatures just to see how they work." Noonan agrees, "If your experiment succeeds and you generate a Neanderthal who talks, you have violated every ethical rule we have," he says, "and if your experiment fails...well. It's a lose-lose." Other scientists think there may be circumstances that could justify Neanderthal cloning.
"If we could really do it and we know we are doing it right, I'm actually for it," says Lahn. "Not to understate the problem of that person living in an environment where they might not fit in. So, if we could also create their habitat and create a bunch of them, that would be a different story."
"We could learn a lot more from a living adult Neanderthal than we could from cell cultures," says Church. Special arrangements would have to be made to create a place for a cloned Neanderthal to live and pursue the life he or she would want, he says. The clone would also have to have a peer group, which would mean creating several clones, if not a whole colony. According to Church, studying those Neanderthals, with their consent, would have the potential to cure diseases and save lives. The Neanderthals' differently shaped brains might give them a different way of thinking that would be useful in problem-solving. They would also expand humanity's genetic diversity, helping protect our genus from future extinction. "Just saying 'no' is not necessarily the safest or most moral path," he says. "It is a very risky decision to do nothing."
Hawks believes the barriers to Neanderthal cloning will come down. "We are going to bring back the mammoth...the impetus against doing Neanderthal because it is too weird is going to go away." He doesn't think creating a Neanderthal clone is ethical science, but points out that there are always people who are willing to overlook the ethics. "In the end," Hawks says, "we are going to have a cloned Neanderthal, I'm just sure of it."
This article by Zach Zorich originally appeared in Archeology Magazine and was republished on NextNature.net with permission. Essay image: An artistic recreation of a Neanderthaler women by Adri en Alfons Kennis. Photo: National Geographic.

gcwffaedrq
Muchas gracias. ?Como puedo iniciar sesion?
Muchas gracias. ?Como puedo iniciar sesion?
Posted on
gulnara
Who exactly will teach the clone Neanderthal about how to live, how to socialize in their own way, of which they know nothing? What language to speak? The clone will have genetics of Neanderthal, yes, but not the knowledge and not the culture of Neanderthal. To throw clones into wilderness and say "Farewell!" and start observing- it all will be wrong. What we will observe then is forming of a Tarzan. Even modern human child, if left to grow in a jungle, will lack culture, language, social skills of a modern human. If clones of Neanderthals are created, they have to be raised in modern society, with all possible freedoms of behavior allowed, but they can not be abandoned, because turning them into wolf children will not give us any valuable information about real Neanderthals.
Posted on
BVG
Come now, G. Gratton, when has ethics EVER slowed the march of science? They'll probably end up selling them as cloned slaves to the highest bidders.
Posted on
G. Gratton
There are so many questions. Why are we doing this? Is it human experimentation? Would the cloned Neanderthal have human rights? If cloned in the U.S. would he/she have full rights as a U.S. citizen? Would he/she learn language? Would the organization which cloned the Neanderthal claim ownership of the person? Is that legal? Similar questions could be asked about the cloning of modern humans. Perhaps we should limit experimentation to lower animals until the ethics are sorted out.
Posted on